Metabolism
on-line - the virtual tutorial room
copyright © 2008 - 2015 David A Bender
Muscle weakness and hypoketotic coma on fasting
TFP is a 3 year old girl who was admitted to the Emergency Department of her local hospital in a coma after two days when she had a fever and had been refusing food. She has a history of muscle weakness, and at the time of her admission her muscle tone was very poor - she was described as being "floppy". A blood sample taken on admission gave the following results:
TFP |
reference range (after overnight fast) |
|
| glucose (mmol /L) | 2.1 mmol /L |
3.5 - 5.5 |
| non-esterified fatty acids (mmol /L) | 2.5 mmol /L |
0.8 - 1.4 |
| ketone bodies (mmol /L) | not detectable |
1.0 - 2.5 |
| pH | 7.40 |
7.35 - 7.45 |
| bicarbonate | 23 |
21 - 25 |
| insulin (mU /L) | 3.5 |
4 - 8 |
What do you think was the cause of her coma?
She is profoundly hypoglycaemic, and this is the immediate cause of her coma - there is no evidence of acidosis, and her plasma insulin is appropriate for her hypoglycaemia (i.e. there is no evidence to suggest that her hypoglycaemia is caused by excess insulin secretion).
What is interesting is that she has a high plasma concentration of non-esterified fatty acids, but no ketone bodies are detectable. This suggests a possible defect in either the synthesis of ketone bodies or the oxidation of fatty acids. Her history of muscle weakness suggests that her underlying problem may be an inability to oxidise fatty acids, which form a major fuel for skeletal muscle. (See also the problem on Muscle weakness, heart failure and profound hypoglycaemia in a young girl ).
She recovered after an intravenous infusion of glucose, although her muscle tone and muscle strength were still poor. Again this is suggestive of a defect in fatty acid oxidation.
The pathway of fatty acid oxidation was elucidated at the beginning of the 20th century, although it was not until the middle of the century that the early deductions were confirmed experimentally.
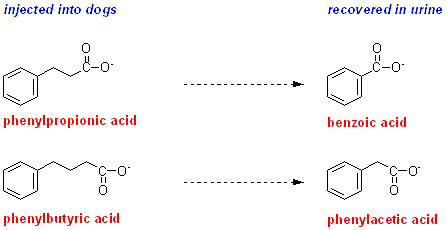
In 1904 Knoop reported the results of injecting dogs with two compounds: phenylpropionic acid and phenylbutyric acid. These are both (very) short chain fatty acids, with a phenyl group in place of the terminal methyl group. Phenylpropionic acid is a phenyl-labelled odd-carbon fatty acid ( 3-carbons), while phenylbutyric acid is a phenyl-labelled even-carbon fatty acid (4 carbons).
The end-products of oxidation of these two fatty acids were excreted as glycine and glucuronic acid conjugates; hydrolysis of the conjugates with hydrochloric acid showed that:
phenylpropionic acid was excreted as benzoic acid conjugates
phenylbutyric acid was excreted as phenylacetic acid conjugates
What conclusions can you draw from these results?
If oxidation of fatty acids occurred as a result of oxidation of one carbon atom at a time to carbon dioxide then you would expect both odd-carbon and even-carbon phenyl-labelled fatty acids to yield the same product, benzoic acid. However, this is not what was observed. The conclusion that Knoop drew was that two carbon atoms are removed at a time in fatty acid oxidation.
Why do you think the term beta-oxidation was adopted for this oxidative removal of two carbon atoms at a time?
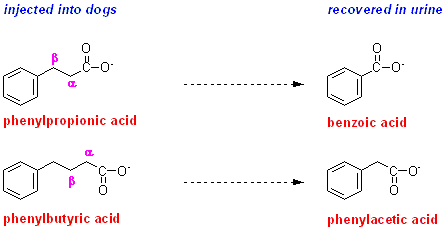 Knoop proposed
that the oxidative attack on the fatty acid must occur at the beta-carbon.
Knoop proposed
that the oxidative attack on the fatty acid must occur at the beta-carbon.
Remember that in this (old-fashioned) nomenclature, the alpha-carbon is the carbon atom to which the functional group for which the compound is named. In this case, the functional group is the carboxyl group. In correct systematic modern nomenclature the alpha-carbon would be carbon-2, since carbon-1 is the carboxyl carbon.
Counting from the alpha carbon, the atoms are then named in (Greek) alphabetical order. We have already seen in the exercise on Fats and oils - are all fats the same? that this nomenclature is still useful, because human beings have enzymes that can introduce additional carbon-carbon double bonds between an existing double bond and the carboxyl group, but not between an existing double bond and the methyl group (the omega-carbon). This means that the position of the first double bond from the omega-carbon determines which family of unsaturated fatty acids a given compound belongs to.
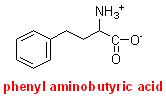 Rats
were fed [14C-U]palmitate (the 16-carbon saturated fatty acid, labelled with
14C in all carbon atoms) and phenyl aminobutyrate, which is excreted as the N-acetylderivative.
Rats
were fed [14C-U]palmitate (the 16-carbon saturated fatty acid, labelled with
14C in all carbon atoms) and phenyl aminobutyrate, which is excreted as the N-acetylderivative.
The N-acetyl phenyl aminobutyrate was recovered from the rat urine, hydrolysed by heating in hydrochloric acid and the acetate was separated by chromatography. It was found that the acetate was labelled with 14C.