Metabolism
on-line - the virtual tutorial room
copyright © 2008 - 2015 David A Bender
Poisoned by unripe ackee fruit
 HG gave a small dinner party, and in honour of one of the guests who was Jamaican,
prepared a traditional Jamaican dish, ackee with rice and salt fish, as well
as a number of other dishes.
HG gave a small dinner party, and in honour of one of the guests who was Jamaican,
prepared a traditional Jamaican dish, ackee with rice and salt fish, as well
as a number of other dishes.
Unfortunately, she had used unripe ackee fruit.
Only two of the guests ate any of ackee dish, and about an hour after the meal both became unconscious. An ambulance was called and they were taken to hospital. Emergency measurement of their blood glucose showed that it was dangerously low: 1.9 mmol /L in one case and 2.1 mmol /L in the other.
What is the normal range of blood glucose?
What emergency treatment should they received?
The normal range of blood glucose is 3.5 - 5.5 mmol /L,
but may rise to 8 mmol /L after a meal.
The obvious emergency treatment would be intravenous glucose
Why does a very low blood glucose concentration lead to loss of consciousness?
The brain is more or less completely reliant on glucose as its metabolic fuel, except in prolonged starvation, and the brain accounts for about 20% of whole body resting energy expenditure. Most of this energy expenditure is involved in transporting sodium and potassium ions across nerve membranes, against their concentration gradient, to maintain electrical activity. The sodium pump (in the brain and all other tissues) accounts for about 22% of whole body resting energy expenditure.
How can sodium and potassium ions be transported across cell membranes against their concentration gradient?
To transport ions against their concentration gradient requires an input of energy.
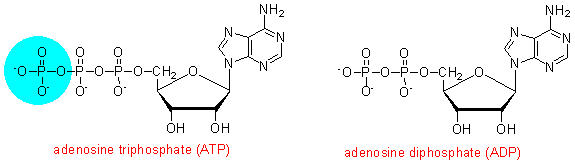
Here we need to introduce the compound adenosine triphosphate (ATP), which has a high free energy of hydrolysis to yield adenosine disphosphate (ADP) and inorganic phosphate. In addition to its role in ion transport, you will see later in this exercise how ATP can be used to drive endothermic reactions in the thermodynamically unfavoured direction, and how it is involved in muscle contraction and hence the performance of physical work.
You will see in later exercises how the phosphorylation of ADP back to ATP is linked to the oxidation of metabolic fuels, so that it acts as an intermediate between the oxidative pathways that are energy yielding and the various processes that are energy requiring.
What happens in sodium / potassium transport is that the carrier in the membrane can exist in two conformations:
With ATP bound it faces inwards, and binds 3 x sodium ions. The binding of sodium ions leads to phosphorylation of the carrier, release of ADP and a conformational switch so that it now faces outwards and expels sodium ions into the extracellullar fluid.
Two potassium ions now bind to the outward facing transporter, causing dephosphorylation. ATP binds to the dephosphorylated transporter, causing it to flip to face inwards again, expelling potassium ions into the cytosol, and ready to bind sodium ions again.
Click here to see more detail of the steps in sodium / potassium transport
Click here to see an animation of this. (Depending on your browser, you may be able to run this animation directly, or it may be downloaded and saved as p-type.exe)