Metabolism
on-line - the virtual tutorial room
copyright © 2008 - 2014 David A Bender
Why does an adult require so much protein in the diet?
It is obvious why a growing child has a relatively high requirement for dietary protein, since there is an increase in the total amount of protein in the body as the child grows.
What is less obvious is why an adult, whose total body protein content does not change, still has a relatively high requirement for dietary protein.
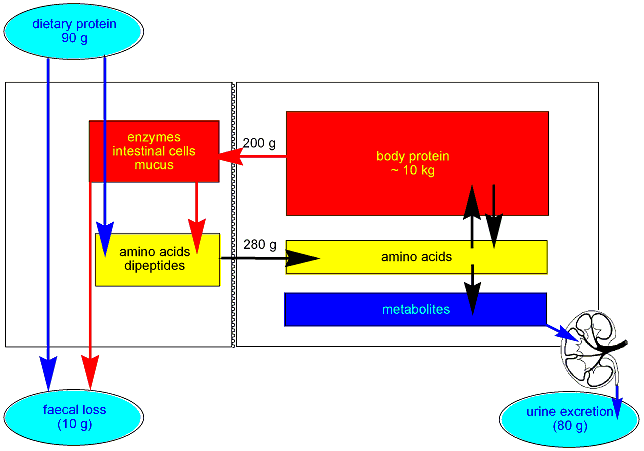
Under normal conditions a healthy adult has an intake of ~90 g of protein per
day, and excretes nitrogenous compounds in urine equivalent to 80 g of protein,
and loses nitrogenous compounds equivalent to 10 g of protein per day in faeces.
This is the state of nitrogen balance or nitrogen equilibrium
intake = output
This simple balance between intake and output hides the fact that some 200 g of protein per day is secreted into the intestinal lumen as digestive enzymes, shed intestinal mucosal cells and the proteins in mucus that protects the intestinal mucosa from the actions of digestive enzymes.
Total daily absorption of amino acids and dipeptides (which are hydrolysed to amino acids during absorption) from the gut is therefore about 280 g.
The amino acids are then used for synthesis of tissue proteins, and a variety of specialised metabolites.
Obviously, in a growing child, in pregnancy, and during recovery from protein losses, there is an increase in the total body protein content, and the output of nitrogenous compounds is less than the intake.
This is positive nitrogen balance - a gain in total body
protein
intake > output
The converse, loss of total body protein is negative nitrogen
balance
output > intake
The graph below shows daily nitrogen balance on a young
man who was fed different amounts of protein for a week at a time, starting
with his habitual intake of 80 g /day, when he was able to maintain nitrogen
balance. 
This was followed by a week with no protein intake, when his balance became very negative.
What conclusions can you draw from the very negative nitrogen balance with zero protein intake?
There is obviously continuing loss of protein, which is not being replaced. There are obviously obligatory losses of nitrogen from the body, meaning that there is a continuing need for a dietary intake of protein.
In the next weeks he was fed 30 g of protein /day for 1 week, then 40 g /day for the second week
What conclusions can you draw from these results?
30 g of protein /day is not enough to meet his requirements, and he is still in negative nitrogen balance, still losing protein from the body. However, at 40 g /day he is able to maintain nitrogen balance, suggesting that this is adequate to meet his requirements and permit replacement of obligatory losses.
He was then fed 100 g of protein /day for 2 weeks. Initially he was in positive nitrogen balance, then after the first week he returned to nitrogen equilibrium.
What conclusions can you draw from these results?
During the first week at 100 g of protein /day he is in positive nitrogen balance, with output less than intake, as he is replacing the body protein that was lost during the weeks with an inadequate intake of protein.
Note that once he has replaced the lost protein he returns to nitrogen equilibrium, with intake = output, and no change in total body protein.
At any level of intake above requirements he is in nitrogen equilibrium - as intake increases, so output of nitrogenous compounds also increases to match the intake.
The table below shows the results of experiments performed by Schoenheimer and colleagues in the 1940s, when he fed rats diet containing an adequate amount of protein for them to maintain nitrogen equilibrium, but labelled with 15N amino acids. Since they were in nitrogen equilibrium, they expected to recover more or less all of the labelled nitrogen in urine and faeces.
| % of label recovered: | [15N]leucine |
[15N]glycine |
| urine | 2.2 |
2.6 |
| faeces | 27.4 |
40.8 |
| non-protein nitrogenous compounds in the body | 8.2 |
11.1 |
| body protein | 56.5 |
44.3 |
| total | 94.3 |
98.8 |
From data reported by Schoenheimer, R 1946. The Dynamic State of the Body Constituents, Cambridge Mass, Harvard University Press.
What conclusions can you draw from these results?
The unexpected finding from these experiments was that less than half of the labelled nitrogen was recovered in urine and faeces, and about half had been incorporated into newly synthesised body proteins. While we now know that there is continual synthesis of proteins in the body, at the time Schoenheimer performed the experiments this was not known. He coined the term dynamic equilibrium to describe the phenomenon of breakdown of existing proteins and replacement, with no change in the total body protein content.
This means that we can modify the diagram of nitrogen balance to include not only protein synthesis, but also protein catabolism.
The driving force for this dynamic equilibrium is the catabolism of tissue proteins, and in fact our current understanding was predicted by the French physiologist Magendie in 1829, when he wrote
"All parts of the body of man experience an intimate movement [we would now call this metabolism] that serves both to expel those molecules that can or ought no longer to compose the body and replace them with new ones"
 The table below shows the results of feeding rats with [15N]labeled
amino acids and measuring the label in different proteins over a period of time.
The time taken for the label to half to half its maximum is called the half-life
of that protein.
The table below shows the results of feeding rats with [15N]labeled
amino acids and measuring the label in different proteins over a period of time.
The time taken for the label to half to half its maximum is called the half-life
of that protein.
| protein | half life |
| ornithine decarboxylase | 11 minutes |
| lipoprotein lipase | 1 hour |
| tyrosine transaminase | 1.5 hours |
| phosphoenolpyruvate carboxykinase | 2 hours |
| tryptophan oxygenase | 2 hours |
| HMG CoA reductase | 3 hours |
| glucokinase | 12 hours |
| serum albumin | 3.5 days |
| arginase | 4 - 5 days |
| lactate dehydrogenase | 16 days |
| collagen | 300 days |
Why do you think some enzymes have a very short half-life (minutes to 3 hours) while other enzymes and proteins are much more stable, and have half lives of days (or almost a year in the case of collagen)?
The enzymes with a short half-life are all key regulatory enzymes in metabolic pathways whose synthesis and catabolism are regulated in response to hormones. With a short half-life it is possible to change the amount of the enzyme in cells in a short time. Note that this is changing the amount of enzyme in a cell, and requires a few hours for the hormone effect to be seen, as opposed to changes in the activity of existing enzyme protein in response to hormone action, which is seen within minutes or seconds of the hormone being secreted.
The enzymes with a half-life of several days can be regarded as "house-keeping" enzymes, whose synthesis is not significantly altered in response to hormone action.
Collagen is a structural protein, and except in times of rapid growth, when the skeleton is being remodelled, turns over only very slowly.
The table below summarises a series of experiments in which young men were fed mixtures of amino acids in amounts that were adequate to permit them to maintain nitrogen balance, but with one amino acid at a time removed from the mixture.
| amino acid omitted | result |
| alanine | nitrogen balance |
| arginine | nitrogen balance |
| asparagine | nitrogen balance |
| aspartic acid | nitrogen balance |
| cysteine | nitrogen balance |
| glutamic acid | nitrogen balance |
| glutamine | nitrogen balance |
| glycine | nitrogen balance |
| histidine | negative nitrogen balance |
| isoleucine | negative nitrogen balance |
| leucine | negative nitrogen balance |
| lysine | negative nitrogen balance |
| methionine | negative nitrogen balance |
| phenylalanine | negative nitrogen balance |
| proline | nitrogen balance |
| serine | nitrogen balance |
| threonine | negative nitrogen balance |
| tryptophan | negative nitrogen balance |
| tyrosine | nitrogen balance |
| valine | negative nitrogen balance |
What conclusions can you draw from these results?
If any one of nine amino acids (histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan or valine) is omitted from the diet then it is not possible to maintain nitrogen balance. This suggests that these nine amino acids are dietary essentials that cannot be synthesised in the body, and must be provided in the diet.
The remaining 11 amino acids can be omitted from the diet without affecting nitrogen balance, suggesting that they can be synthesised in the body, and are not dietary essentials. They are generally known as non-essential or indispensable amino acids.
Two of the non-essential amino acids are only synthesised in the body from essential amino acids: cysteine from methionine and tyrosine from phenylalanine. This means that if the essential precursor is not provided in adequate amounts then the non-essential amino acids becomes a dietary requirement. Equally, if the non-essential amino acid is provided in the diet then the requirement for the essential precursor is reduced. This is important with the two sulphur amino acids methionine and cysteine, since most diets are limited by their content of [methionine + cysteine].
Click here to run the computer
simulation program that permits you to determine the requirements for total
protein and individual amino acids by measuring nitrogen balance.
(Some browsers will allow you to run the program directly, others may require
you to download and save the program before running it)
Only three of the non-essential amino acids can be regarded as completely dispensable, since they are synthesised from common metabolic intermediates:
alanine is synthesised from pyruvate
aspartic acid is synthesised from oxaloacetate
glutamic acid is synthesised from alpha-ketoglutarate
The capacity to synthesise other non-essential amino acids may be inadequate to meet requirements at times of high demand. For example:
glutamine in response to surgical trauma and sepsis
arginine at times of high protein intake or rapid growth
glycine with high intakes of some xenobiotics which are excreted as glycine
conjugates, and in rapid growth because of the requirement for collagen synthesis
proline in severe trauma because of the requirement for collagen synthesis
Protein losses in response to trauma
In response to physical trauma of various kinds, and fever, there are considerable losses of tissue protein. The catabolic loss may be 6 - 7% of total body protein over 10 days. The table below shows these losses over 10 days in various conditions.
tissue loss |
blood loss |
catabolism |
total |
|
| fracture of femur | - |
200 g |
700 g |
900 g |
| muscle wound | 500 - 750 g |
150 - 400 g |
750 g |
1350 - 1900 g |
| 35% burns | 500 g |
150 - 400 g |
750 g |
1400 - 1650 g |
| gastrectomy | 20 - 180 g |
20 - 100 g |
625 - 750 g |
645 - 850 g |
| typhoid fever | - |
- |
675 g |
675 g |
[From data reported by Cuthbertson, DP. Physical injury and its effects on protein metabolism, pp 373-414 in Human Protein Metabolism Vol 2 (Munro HN & Allison JB, Eds), Academic Press, New York, 1964]