Metabolism
on-line - the virtual tutorial room
copyright © 2008 - 2015 David A Bender
Sugars, starches and the glycaemic index
A group of people consumed two test meals, each containing the same amount of total carbohydrate, on two separate occasions. The graph shows the average changes in blood glucose concentration over 4 hours after each meal.

Can you account for the differences?
What would you expect to see if the subjects had consumed an equivalent amount of glucose?
The carbohydrate in meal 2 is obviously more rapidly and completely digested than that in meal 1.
Glucose does not require digestion, and is rapidly absorbed, so the response of blood glucose to a test dose of glucose would be similar to that after test meal 1 - or perhaps a higher peak achieved slightly more rapidly.
What is meant by the term glycaemic index?
The glycaemic index of a food is the extent to which it raises the blood concentration of glucose (i.e. the area under the curve in the graph shown above), compared with that for a test dose of the equivalent amount of glucose or a standard carbohydrate food that is rapidly digested, such as freshly boiled mashed potato or white bread.
Would you expect to see different results for measurement of serum insulin
after these two test meals?
If so, why?
If not, why not?
 Insulin
is secreted in response to increasing blood glucose concentrations, so you would
indeed expect to see a lower concentration of serum insulin at each time point
after meal 1, which causes less increase in blood glucose, than after meal 2.
Insulin
is secreted in response to increasing blood glucose concentrations, so you would
indeed expect to see a lower concentration of serum insulin at each time point
after meal 1, which causes less increase in blood glucose, than after meal 2.
Sugars, starches and the glycaemic index
How can you classify dietary carbohydrates according to their chemistry?
 We can
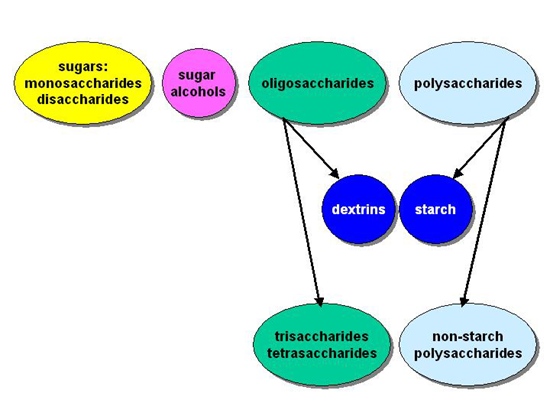
classify carbohydrates into three main groups:
We can
classify carbohydrates into three main groups:
the sugars (mono- and disaccharides),
oligosaccharides (polymers of 3 - 10 monosaccharide units)
polysaccharides (polymers of several hundred monosaccharide units).
It is conventional to consider the sugar alcohols separately from the simple sugars.
The nutritionally and metabolically important monosaccharides are glucose, galactose and fructose:

How does galactose differ from glucose?
Galactose differs from glucose in the orientation of the hydroxyl group at carbon-4.
How does fructose differ from glucose?
Fructose has a keto group at carbon-2 (and hence is a ketose), rather than an aldehyde group at carbon-1, as in glucose and galactose (which are aldoses). This means that while glucose and galactose are chemically reducing compounds, fructose is not. It also means that when ring closure occurs, glucose and galactose form 6-member pyranose rings, while fructose forms a 5-member furanose ring.
 Glucose,
galactose and fructose are all 6-carbon sugars (hexoses). Two 5-carbon sugars
(pentoses) are biochemically important: ribose and dexxyribose.
Glucose,
galactose and fructose are all 6-carbon sugars (hexoses). Two 5-carbon sugars
(pentoses) are biochemically important: ribose and dexxyribose.
The sugar alcohols are formed by reduction of the aldehyde or keto group of a sugar to a hydroxyl group, and therefore are often called polyols. For example, the sugar alcohol derived from glucose is sorbitol:
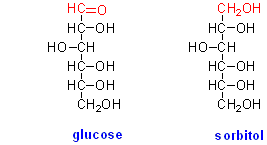
What are the major dietary disaccharides?
The main dietary disaccharides are sucrose, lactose, trehalose, maltose and isomaltose:

How could you investigate how monosaccharides and sugar alcohols are absorbed from the small intestine?
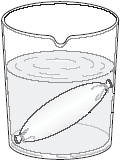 The classical
technique is the "everted gut sac". The small intestine of a rat or
other experimental animal is dissected out, washed with an appropriate buffer
solution, then everted - turned inside out by rolling it over a metal rod. Small
sections of the everted gut (4 - 6 cm long) are then tied off at one end, filled
with buffer solution and tied off at the other end. They are then suspended
in a buffer solution containing the compound of interest, as well as a small
amount of glucose to permit them to metabolise. The contents of the sac represent
what has been transported from the mucosal surface of the gut (which now faces
outwards into the incubation medium). This means that sampling the contents
of the sac after an appropriate time of incubation permits you to measure what
has been transported.
The classical
technique is the "everted gut sac". The small intestine of a rat or
other experimental animal is dissected out, washed with an appropriate buffer
solution, then everted - turned inside out by rolling it over a metal rod. Small
sections of the everted gut (4 - 6 cm long) are then tied off at one end, filled
with buffer solution and tied off at the other end. They are then suspended
in a buffer solution containing the compound of interest, as well as a small
amount of glucose to permit them to metabolise. The contents of the sac represent
what has been transported from the mucosal surface of the gut (which now faces
outwards into the incubation medium). This means that sampling the contents
of the sac after an appropriate time of incubation permits you to measure what
has been transported.
What would you conclude if the concentration of a sugar inside the sac was greater than that outside?
If there is a higher concentration of a sugar inside the sac than outside then this must be active transport - directly or indirectly linked to utilisation of ATP. Both glucose and galactose are actively transported across the intestinal mucosa, linked to the transport of sodium ions down their concentration gradient. In turn, the sodium gradient has been set up linked to the hydrolysis of ATP to ADP and phosphate.
In a series of experiments it was found that when both glucose and galactose were present in the incubation medium, they competed with each other - the more galactose was present, the less glucose was absorbed. Similarly, the more glucose was present, the less galactose was absorbed.
What conclusion can you draw from this?
If glucose and galactose compete with each other then they must be transported by the same carrier. The total amount of sugar (glucose + galactose) transported will be more or less constant as the proportions of each change. If they were transported by separate carriers then there would not be competition, but rather when both sugars were present in the incubation there would be increased total sugar transport.
Fructose, sugar alcohols and various other monosaccharides are absorbed by carrier-mediated diffusion, and so cannot be transported against their concentration gradient.
What do you think is the likely result of ingesting a relatively large amount of fructose or sugar alcohol?
A relatively large amount of fructose or sugar alcohol exceeds the capacity to absorb it. This leaves a relatively high concentration of sugar (or sugar alcohol) in the intestinal lumen. This has an osmotic effect, drawing water into the intestinal lumen. It also provides a substrate for fermentation by intestinal bacteria to 2- and 3-carbon acids, so increasing yet further the osmotic effect. The net result of this is an osmotic diarrhoea - diarrhoea caused by the relatively large amount of water drawn into the intestinal lumen by the unabsorbed sugar.